Gabapentin is a pharmaceutical drug that has a white solid (small crystals) physical appearance. The molecular formula of gabapentin is C9H17NO2, and it has a…
What is Gabapentin ? Gabapentin capsules, tablets, and oral solution are used along with other medications to help control certain types of seizures in people…
What is Gabapentin ? Gabapentin is used to help control partial seizures (convulsions) in the treatment of epilepsy. This medicine cannot cure epilepsy and will…
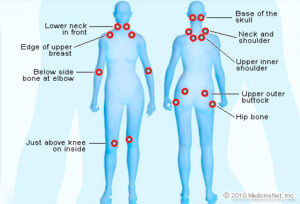
The symptoms of fibromyalgia include pain and tenderness throughout the body that is often associated with other conditions that reduce a person’s ability to function…
In an Research, Patients were started on one 300-mg capsule of gabapentin or matching placebo, and then were titrated weekly from 900 mg/day (end of week…
What is Fibromyalgia? Fibromyalgia (fi·bro·my·al·gi·a) is a condition that causes pain all over the body (also referred to as widespread pain), sleep problems, fatigue, and…
What is gabapentin? Gabapentin is a prescription drug. It comes as an oral capsule, an immediate-release oral tablet, an extended-release oral tablet, and an oral…
What your doctors and your pharmacists want to know about your health questionnaires when you buy Gabapentin, buy fioricet online. The Doctors and pharmacists need…
What is gabapentin? Gabapentin is an anti-epileptic drug, also called an anticonvulsant. It affects chemicals and nerves in the body that are involved in the cause of…
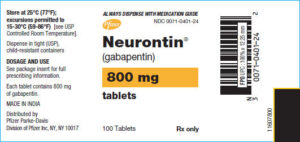
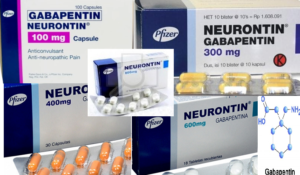
Gabapentin capsules are available under different trade names and at different strengths. Gabapentin is used most commonly to treat epilepsy and neuropathic, or nerve, pain. The trade names…